Sumitomo Heavy Industries, Ltd. has started to a comprehensive review of the SAKIGAKE Designation System in order to speedily evaluate the efficacy of accelerator based BNCT systems in Japan.
May 20, 2019
Sumitomo Heavy Industries, Ltd. (President: Shinji Shimomura) has performed the phase II trial of the BNCT*1) (Boron Neutron Capture Therapy) system using an accelerator (cyclotron), targeting carcinoma of the head and neck region in collaboration with STELLA PHARMA CORPORATION (President: Tomoyuki Asano). These trials were performed to verify the safety and effectiveness of BNCT. The evaluation for the response rate (tumor reduction effect) on 21 subjects*2) as of 90 days after the completion of the trials has been completed, and it has been confirmed that the anticipated results were achieved. In response to this, Sumitomo Heavy Industries, Ltd has started comprehensive review utilizing the SAKIGAKE Designation System*3) in preparation for the manufacture and sales approval of the world’s first BNCT system. While the initial goal of the BNCT system targets carcinoma of the head and neck region, based on results of the above evaluation, clinical trial aiming to expand applications/indications in the near future is simultaneously in progress.
The details of this trial result are scheduled to be presented at the 2019 ASCO ANNUAL MEETING*4), which will be held in Chicago, Illinois, in the U.S. in May of this year.
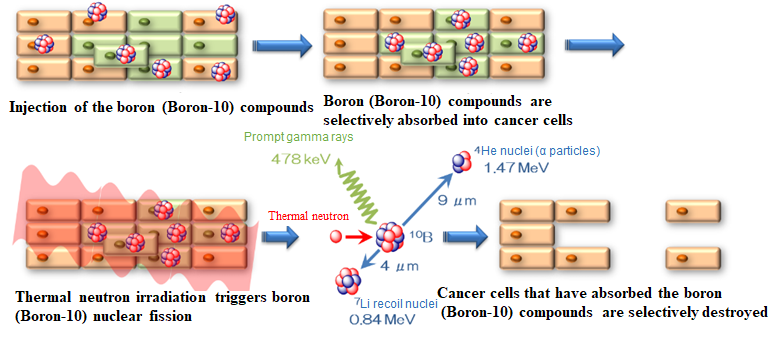
*1 BNCT is a type of cancer radiotherapy. In this therapy method, a boron medical agent for BNCT is given to cancer patients whereby cancer cells selectively absorb boron (Boron-10). The results is that low-energy neutrons are radiated which in turn causes fission of the boron (Boron-10) nuclei inside the body. This reaction releases α particles (helium nuclei) and Li recoil nuclei (lithium nuclei), which possess enough energy to damage cancer cells. The range of these charged particles is approximately 9μm and 5μm respectively. This range is approximately the size of a cancer cell. This makes it possible to selectively destroy cancer cells, which have absorbed the boron (Boron-10), while minimizing damage to surrounding healthy cells.
*2 Subjects are those with unresectable locally-recurrent head and neck carcinoma and unresectable locally-advanced non-squamous-cell carcinoma in the head and neck region.
*3 SAKIGAKE Designation System was implemented in 2015 as a strategic package supported by the Japanese government to speedily promote practical applications of innovative pharmaceuticals, medical-devices and regenerative medicine products for life-threatening diseases in Japan. The goal is commercial implementation of this treatment method before it is available globally by covering the entire process, including research, clinical trials, safety responses, insurance applications and international deployment. This project was specified as one of the subjects for SAKIGAKE Designation System on February 29, 2017, under the name of “Boron Neutron Capture Therapy (BNCT) system”.
*4 American Society of Clinical Oncology (ASCO) is the world’s largest cancer society. The annual meeting of 2019 will be started from May 31 in Cicago, IL.